Contributing Experts at Medrio: Melissa Newara, VP of Subject Matter Expertise, and Tina Caruana, eClinical Solutions (Digital & Decentralized Trials) Subject Matter Expert
Real world data (RWD) and real world evidence (RWE) have great research implications and financial potential. Sponsors are tapping into electronic health records (EHRs), questionnaires, claims, genomics, wearable, and beyond to gather more real world data and gain new insights.
Between 2004 and 2022, the volume of studies using RWD grew by almost 2,000 percent. But as clinical trial sponsors work to incorporate more RWD and RWE into research studies—particularly Phase IV—they face both challenges and opportunities.
With insights from Medrio experts Tina Caruana, eClinical Solutions Subject Matter Expert, and Melissa Newara, VP of Subject Matter Expertise, this blog covers:
- Differences between real-world data and real-world evidence
- Challenges of RWD capture for Phase IV trials
- Benefits of front-end digital processes
To explore how to design an efficient, quality Phase IV trial that integrates back-end data management with a seamless upfront experience, read our eBook.
The Definitions of Real World Data and Real World Evidence
In its December 2018 framework for RWE, the Food and Drug Administration (FDA) defined RWD and RWE plainly:
- Real World Data (RWD) is data coming from non-trial sources about patient outcomes and care delivery.
- Real World Evidence (RWE) is the clinical evidence generated by analyzing that RWD.
Another way to put it: RWD is the how, and RWE is the why.
Increasingly, more people are understanding the value of both inputs, especially as technology makes this data more possible. In a 2022 Deloitte survey, more than seven in 10 people said they could now generate RWE more efficiently thanks to digital tools such as visualizations and analysis software.
In an article for PMLiVE in early 2023, Aneta Tyszkiewicz, Associate Director, Digital & Data, European Federation of Pharmaceutical Industries and Associations (EFPIA), emphasized the importance of real-world data and real-world evidence.
“The main advantage over data and evidence from clinical trials is that RWD and the resulting RWE reflect real clinical practice. RWD can enable a better view of the actual effectiveness and characteristics of a medical product or technology, and create new insights to improve health outcomes for patients and efficiency of healthcare systems,” said Aneta.
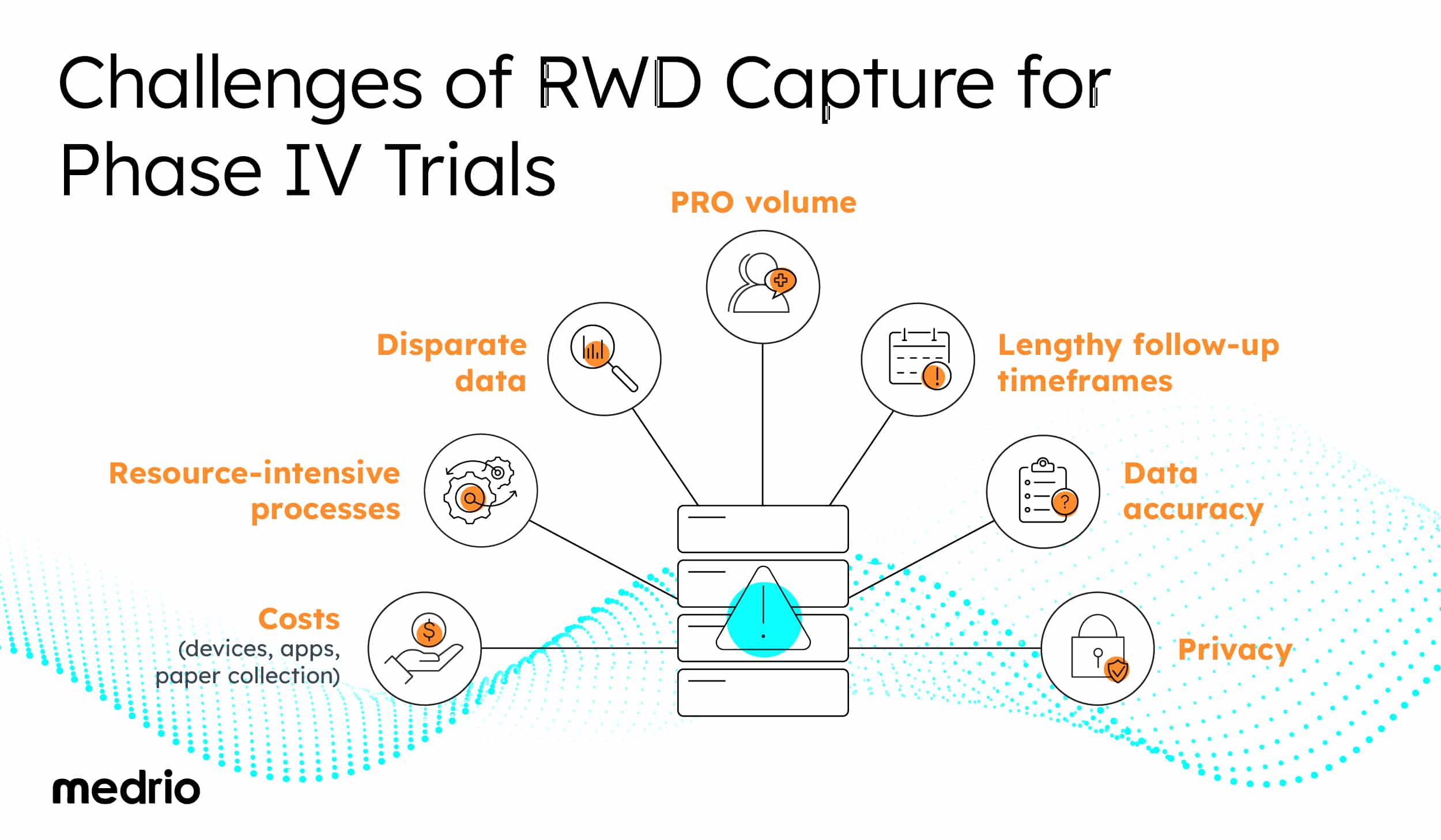
The Challenges of RWD Capture for Phase IV Trials
With all this data available, sponsors are understandably eager to use it. From 2020 to 2022, the proportion of Phase IV trials using insights from the real world more than doubled.
But the size and scope of Phase IV studies, along with the rapid uptake of digital tools to collect RWD, have brought growing pains.
Challenges with RWD capture include:
- Disparate and complicated data
- Large volumes of patient-reported outcomes
- Lengthy follow-up timeframes
- Legibility and accuracy of patient-reported data
Large volumes of data from patient-reported outcomes
The sheer size of Phase IV studies—sometimes thousands of participants—can create barriers to directly capturing input from patients.
These barriers worsen when it comes to manually consolidating data points for actionability, especially when conducting a long-term follow-up.
Resource-intensive processes
Reliance on traditional data collection methods often makes RWD collection costly and resource-intensive.
Some common costly and resource-intensive methods include:
- Collecting paper-based information
- Managing apps
- Providing devices
Sponsors may underestimate the time involved in consolidating paper forms into trial execution workflows. According to Tina, “It’s very complicated and frequently prone to errors, such as duplications, missing data, and the need for data transcription into the [Clinical Data Management System] CDMS.”
Also Read: ePRO vs Paper Strategies in Clinical Trials
Legibility concerns
While much RWD comes from wearables and other digital devices, some sponsors also collect it from paper forms. This approach, however, comes with challenges.
For example, a recent survey by MedTech Dive and Medrio of 150 respondents involved with medical device trials found that 43 percent of people considered illegible or extraneous data to be their biggest challenge with paper logs.
For example, paper questionnaires may contain:
- Illegible handwriting
- Misplaced text in the margins
- Missing entire pages
There’s also the so-called “parking lot syndrome” in which participants hastily fill out their questionnaires just minutes before their scheduled visit.
These paper-based issues can drive up costs and slow down study timelines. Of note, paper logs don’t always help enhance the value those instruments were designed to deliver, explains Tina.
To explore how to overcome these challenges, read our eBook.
The Benefits of Front-End Digital Processes
Sponsors generally know they need to operationalize trial functions. Many have already done so on the back end through electronic data capture (EDC) within clinical data management systems (CDMS).
But while steps such as EDC help studies function at a Phase IV scale, they’re not the only processes worth implementing.
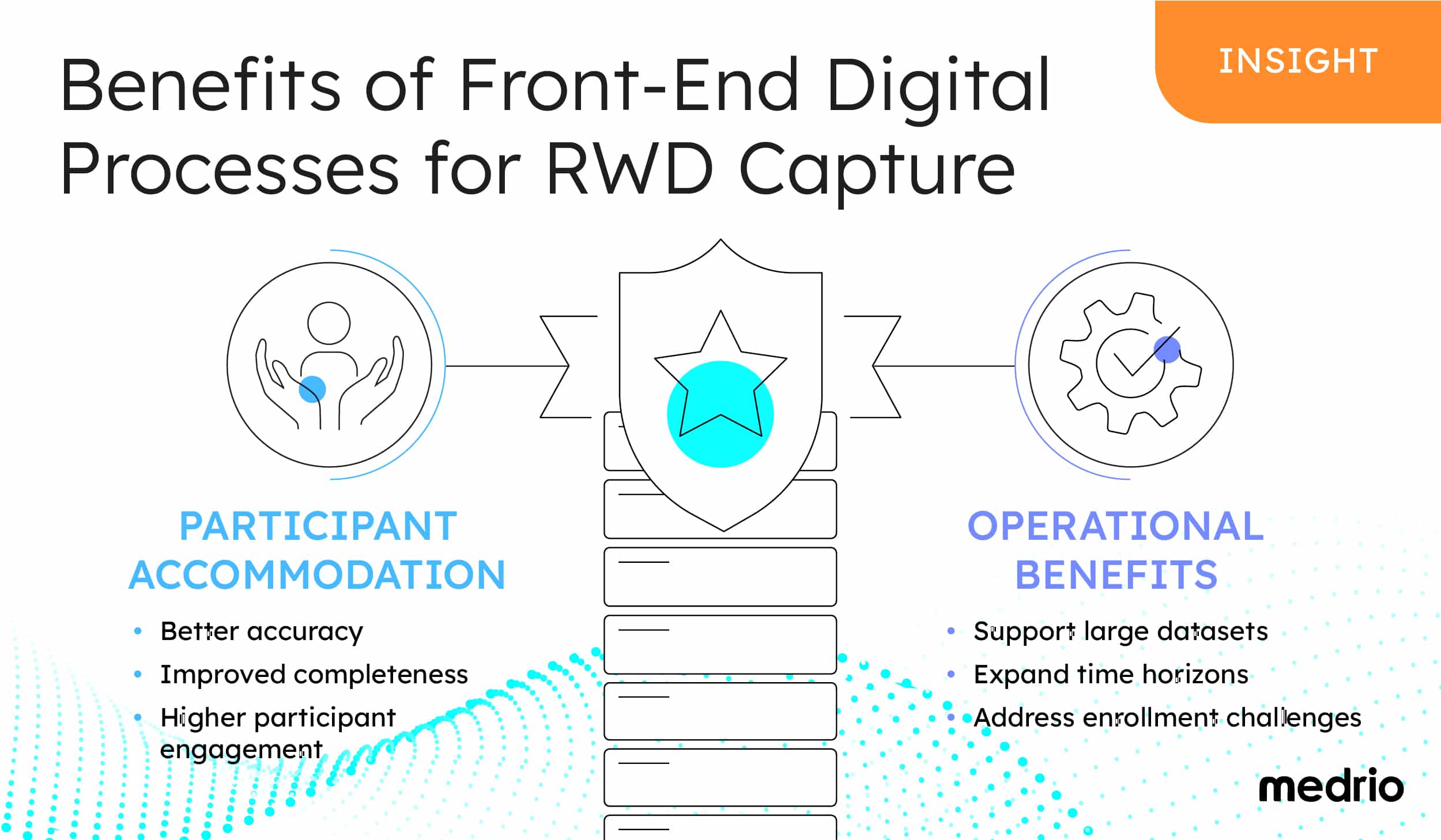
By focusing on the optimization of the front-end experience, sponsors can:
- Support greater participant engagement
- Yield significant operational benefits
Participant accommodation
Participants want and need to control their participation in clinical research. Melissa emphasizes, “With digital activities, they [patients] can participate in ways that are most comfortable for them without having to carry around paper assessments or be forced into specific routines that aren’t feasible.”
By investing in a purpose-built, secure, and integrated technology for activities such as registration, consent, and engagement, sponsors can accommodate participants —instead of expecting participants to accommodate the trial.
Accommodating patients leads to greater:
- Accuracy
- Completeness
- Engagement
Melissa explains, “Flexibility and patient centricity are all about figuring out how to fit into people’s everyday lives to ensure that they’re providing honest, accurate data.”
Operational benefits
Sponsors also glean operational benefits from RWE—from cost and resource savings to potentially better real-world data that’s more easily integrated into the Phase IV study and turned into real-world evidence.
“We’re seeing more and more sponsors seeking patient-facing technologies, such as ePRO and eConsent, to help capture the growing volumes of participant data in more accurate and efficient ways,” Tina says.
By optimizing front-end digital processes, sponsors can:
- Support large datasets
- Expand time horizons
- Address enrollment challenges
Support large datasets. Study teams can set up the data cleaning and standards that make that data more actionable and relevant to the study.
Expand time horizons. If participants have an accessible digital tool that engages them in the study long-term, 10- to 15-year safety follow-up schedules may be more achievable. These follow-ups also can be more scalable and sustainable across large Phase IV study populations.
Address enrollment challenges: By decentralizing participant-facing instruments for enrollment activities, sponsors offer participants greater autonomy. This may help sponsors overcome systemic under-enrollment problems due to limited site availability or clinical research coordinator bandwidth.
Melissa adds, “The value there is not necessarily in the processing of patients who are already enrolled, but in getting more diverse, larger groups of participants to engage in the study without always needing a centralized site or multiple sites. You can promote self-recruitment and self-enrollment in new and creative ways that don’t have to be reliant on traditional enrollment pathways.”
The Future of Real World Evidence
Real-world evidence will become ever more important as sponsors work to elevate the participant’s voice in clinical research. As RWE’s financial and research value becomes more clear, sponsors overcome challenges by incorporating front-end digital processes that set them up for long-term success.
Want to know more? Find how to design an efficient, quality Phase IV trial that delivers the best of both worlds—back-end data management integrated with a seamless upfront experience.
Download our eBook – “Real-World Evidence and Phase IV: Making Enrollment and Engagement Work at Scale.”



