Author: Nicole DuPont Latimer, CEO of Medrio
We all experience illness at some point in life, which is why clinical trials are essential. Clinical trials help life science organizations ethically bring medicine to market, giving us better ways to alleviate symptoms and recover from illness.
Through exploring the use of digital data capture in clinical trials, an alarming truth remains; many life sciences companies are not fully operationalizing their stated commitment to helping patients and participants.
Seeing Through the Double Standard
When analyzing 35 of the globe’s highest-grossing life sciences companies, a striking 83% included the health and safety of patients in their mission statements. On average, these companies referred to patients 2.5 times in each mission statement.
These references indicate the strong focus and commitment to prioritizing patient well-being. However, many studies often still use outdated data collection methods like manual patient surveys and paper forms.
For example, a clinical trial consulting firm, DT Consulting, surveyed sites conducting more than 3,000 interventional trials and discovered:
- Only 75% of life sciences companies use or plan to use electronic data capture (EDC) to record patient data in their clinical trials
- Less than 50% of trials use or plan to use electronic patient-reported outcomes (ePRO) and eConsent
- Less than 25% of sites use or plan to use other electronic data sources (eSource) to capture data digitally during their trials

By not implementing digital data sources in clinical trials, many of these companies have yet to put their patient-centric philosophies fully into practice in clinical trials. Reliance on traditional approaches can burden patients and lead to lower-quality data, potentially compromising patient care.
Modern technology can support patient-centric trials and bring a life science company’s values to fruition. Data capture through digital solutions also allows for remote data capture, providing flexible options for patient participation. It’s time for these organizations to adopt digital data capture methods in clinical trials to uphold their commitment to ethical standards and improve the patient experience.
Bridging the Gap
Capturing clinical data digitally directly or through devices, wearables, or other sources rids off the hassles of traditional paper-based methods and creates efficiencies in trial processes.
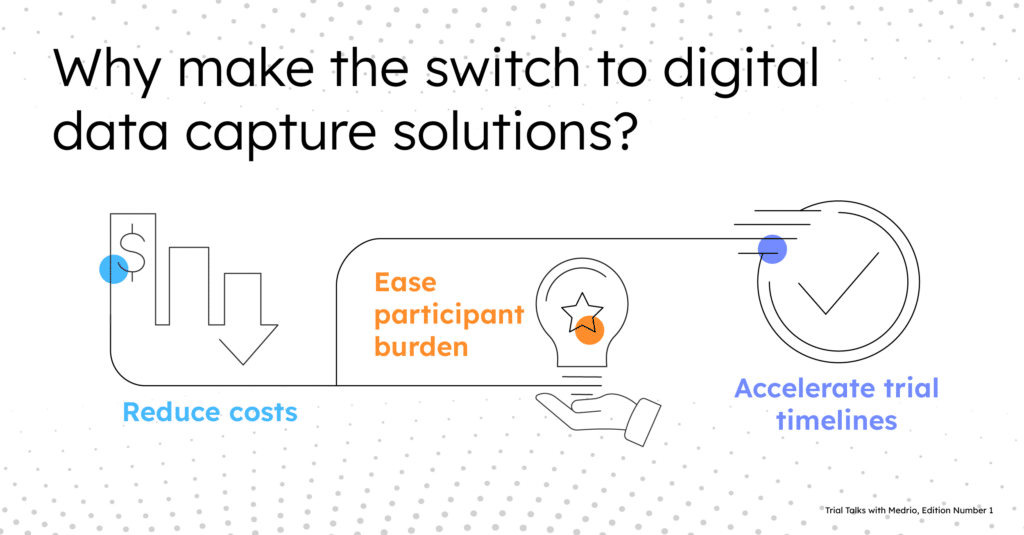
Why make the switch to digital data capture solutions?
- Reduce costs: Sponsors experience financial benefits by decreasing the time from data collection to database lock. It also can potentially remove source data verification (SDV) processes.
- Ease burden: Participants experience greater efficiency by reducing travel time to the research site with the potential of remote data capture options.
- Accelerate timelines: CROs and sponsors experience swift data transfer, which removes any delay caused by intermediaries and allows for timely decision-making.
Also Read: ePRO vs Paper Strategies in Clinical Trials
Implementing digital data capture boosts efficiency, making trial timelines faster and getting treatments to patients sooner. Companies save money, which means more therapies can be developed to benefit even more patients.
But efficiency isn’t the only benefit of digital data capture.
How to Choose the Right eClinical Technology to Power Your Studies
If you want to discover the right eClinical technology for your study, read our eBook that explores different digital tools that improve the participant experience.
Ensuring Quality, Accurate Data
Going digital also allows for more accurate data capture. The direct entry of data into digital systems improves data accuracy. Further, digital data entry helps to remove potential errors that can occur when transferring data from paper.
How does digital data capture improve data quality and accuracy?
- Directly transferring digital data to a centralized database eliminates errors from paper-based intermediaries.
- Passive data collection, like through wearable devices, requires no input from patients, reducing opportunities for human error.
- Electronic checks ensure accurate data capture by identifying and notifying of mistakes in data fields.

The heightened data accuracy, once again, accelerates clinical trials, ultimately benefiting patient health. Additionally, researchers can process data more effectively, enhancing overall patient safety throughout the clinical trial.
Digitally-captured data is also easier to analyze. Audit trails of electronic transfers show researchers exactly where it is coming from. In addition, real-time remote data monitoring allows data to be collected more frequently, thus minimizing data gaps.
All of these factors combine to enhance patient safety because researchers can more easily detect unusual data, identify the source, and intervene with patients more rapidly.
Success Story: Atlantic Research Group Builds Competitive Advantage with Medrio EDC
Choosing Digital Data Capture for Patient-Centric Clinical Trials
When considering the many benefits of digital data capture, it becomes clear that it’s a more patient-friendly approach than traditional paper-based methods.
Participants gain several advantages: fewer site visits, timely interventions facilitated by increased data accessibility, more accurate data for regulatory approval, and cost reductions enabling further drug discovery.
The mission statements of most life sciences companies emphasize patient health and well-being. However, if patient priorities are truly important, why is digital data capture, such as EDC, ePRO, eConsent and other methods of collecting eSource not used more extensively in their clinical trials?
To genuinely fulfill their patient-centric missions, life sciences companies should provide digital data capture options in their clinical trials. It’s time to align words with actions for the benefit of patients.
How to Choose the Right eClinical Technology to Power Your Studies
If you want to discover the right eClinical technology for your study, read our eBook that explores different digital tools that improve the participant experience.



