Contributing Experts at Medrio: Ian Davison, RTSM Subject Matter Expert
The primary goal of a clinical trial supply strategy is to ensure the right product is available and dispensed to the right participant at the right time. Second, come the goals of increasing efficiency and reducing waste. To reach these goals, sponsors need to do more than just manage their supply chain, they need supply chain optimization.
At the end of the study, many sponsors must destroy unused stock. This inefficient process ultimately wastes costly investigational product (IP) and adds to the expense of running a clinical trial. Meanwhile, the entire product life cycle must be tracked, including its return or destruction, adding to the management burden necessary to complete a trial.
When sponsors can develop a lean, streamlined supply chain operations strategy, they alleviate staff workload and significantly reduce IP waste. Developing this strategy is easier said than done, but with the right partners, an optimized supply chain is feasible and cost-effective.
In this article, we answer the questions:
- When should you think about supply chain logistics?
- How do you plan for study complexity?
- How can you make best use of staff resources?
- Are you optimizing your supply chain?
Learn more about how to optimize your supply chain in this downloadable guide for clinical trial supply chain optimization.
When Should You Think About Supply Chain Logistics?
Planning a clinical trial involves so many critical moving parts, that it’s tempting to de-prioritize supply chain logistics.
However, it’s important for sponsors and CROs to ask supply chain questions as early in the clinical trial planning process as possible. Supply chain optimization has to be considered during the protocol writing phase so the dispensing plan is amenable to other components of the clinical trial and the right trial decisions can be made at the right time.
Supporting a coordinated and participant-focused dispensing plan involves aligning manufacturing, packaging, labeling, and demand-driven logistics with the dispensing plan. This alignment ensures efficient inventory use while safely minimizing waste.
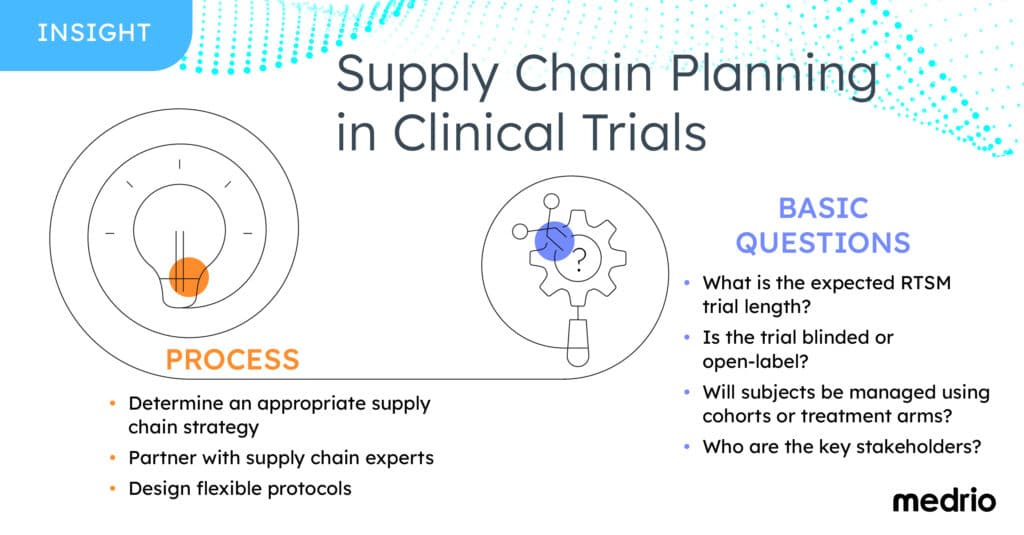
Determining an Appropriate Supply Chain Strategy
Delays in therapeutic supply stall study timelines, but ordering too much IP only to destroy it later wastes resources. Sponsors and CROs who formulate their supply chain approach early in the planning process create a smoother, more streamlined process that reduces waste and keeps studies running on time.
Determining the right strategy first depends on the individual therapeutic. There is no one-size-fits-all solution to supply chain optimization.
For example, if the therapeutic is relatively inexpensive to manufacture and is shelf-stable, sponsors can worry less about potential waste and elaborate logistics. Sponsors can send a big product shipment at site activation and send additional product if supplies run low.
But when the therapeutic expires quickly, is in short supply, is very costly, or is a controlled substance, a more demand-driven or potentially “just in time” approach that supplies small quantities of the product when needed is more appropriate.
Packaging is another area that should be considered. How a drug is packaged determines its flexibility.
For example, say a participant receives eight-week treatments, dispensing every two weeks. If the product is packaged per participant, the investigator must initially open the entire eight-week supply. But if the participant later drops out, any remaining product in the box must be discarded. This approach is essentially designing waste into the system.
Instead, if the drug were packaged in smaller units and dispensed at the appropriate times, a participant dropping out would mean no product loss. Packaged separately, the doses intended for that participant could be given to other participants, instead.
This scenario highlights how one aspect of the supply chain, such as packaging, has a domino effect on other aspects, such as dispensing, which is determined by the protocol. Because the protocol directives affect the supply chain in myriad ways, considering the entire drug life cycle during the clinical trial planning process helps prevent future problems or added expenses.
Partnering with Supply Chain Experts
A randomization and trial supply management (RTSM) system accompanied by an expert, consultative team can help to provide solutions to optimize supply chain processes.
In the above scenario, an RTSM can recognize the remaining doses as available and dynamically allocate them in a blinded manner to any eligible participant who needs them, keeping viable IPs out of the waste-and-destruction cycle.
An accompanying expert RTSM team can also consult with sponsors and CROs early in the study design process to anticipate and plan for scenarios like these. Outside experts leverage their supply chain optimization experience to suggest modifying packaging and dispensing plans, providing greater flexibility and reducing future waste while centering participants’ needs
Plus, it’s important to partner with an RTSM system that is designed with these scenarios in mind and can adapt as enrollment changes or to accommodate mid-study protocol amendments.
Designing Flexible Protocols
Packaging and dispensing plans must fit the protocol, but building flexibility into the protocol from the beginning, such as allowing unused product to be assigned to other participants when appropriate, can reduce delays and added costs.
For instance, complex participant packs with multiple products in each pack cover multiple visits. If everything goes as planned, complex packaging can work well. This approach migrates complexity from the clinic to the packaging and labeling group.
However, trials rarely, if ever, proceed exactly as planned.
IP kits can be spoiled due to temperature excursions, or vials can be dropped or broken. Participants drop out or remove consent, or protocols change based on findings within the trial. You can’t go back in time and repackage. Awareness of potential problems and changes early in the planning stage is essential to creating a flexible, risk-based plan with resilience built-in.
A consultative RTSM partner is the glue that pulls these disparate processes together and examines the intersection of inventory, dispensing schedules, packaging and labeling, and distribution systems. They begin with the end goal: to ensure that the right drug reaches the right participant while avoiding waste as much as possible.
How Do You Plan For Complexity?
Clinical trials are increasingly complex. No two clinical trials are the same, and each therapeutic brings challenges.
Many biopharmaceuticals have a short shelf life and require precise temperature tracking to avoid spoilage. Other drugs, such as controlled substances, are shelf-stable but highly regulated and cannot be stored in large quantities, requiring multiple shipments. Cutting-edge IP is often highly expensive and is best produced in small quantities to avoid wasted product.
Likewise, shipping costs vary wildly from trial to trial. Temperature-controlled shipping, packaging options, shipment timing, and potential stockouts create logistical headaches.
These costs cannot be eliminated, but they can be mitigated. The drug must be tracked and logged at every stage, from manufacturing to storage to participant compliance, amassing a multitude of data. However, many companies rely on multiple, siloed processes to manage the above, increasing costs and study timelines.
Increasingly, companies are turning to single, unified platforms that eliminate these silos and manage the entire supply chain from end to end, using automation and cloud-based systems to improve an often overly complicated system.
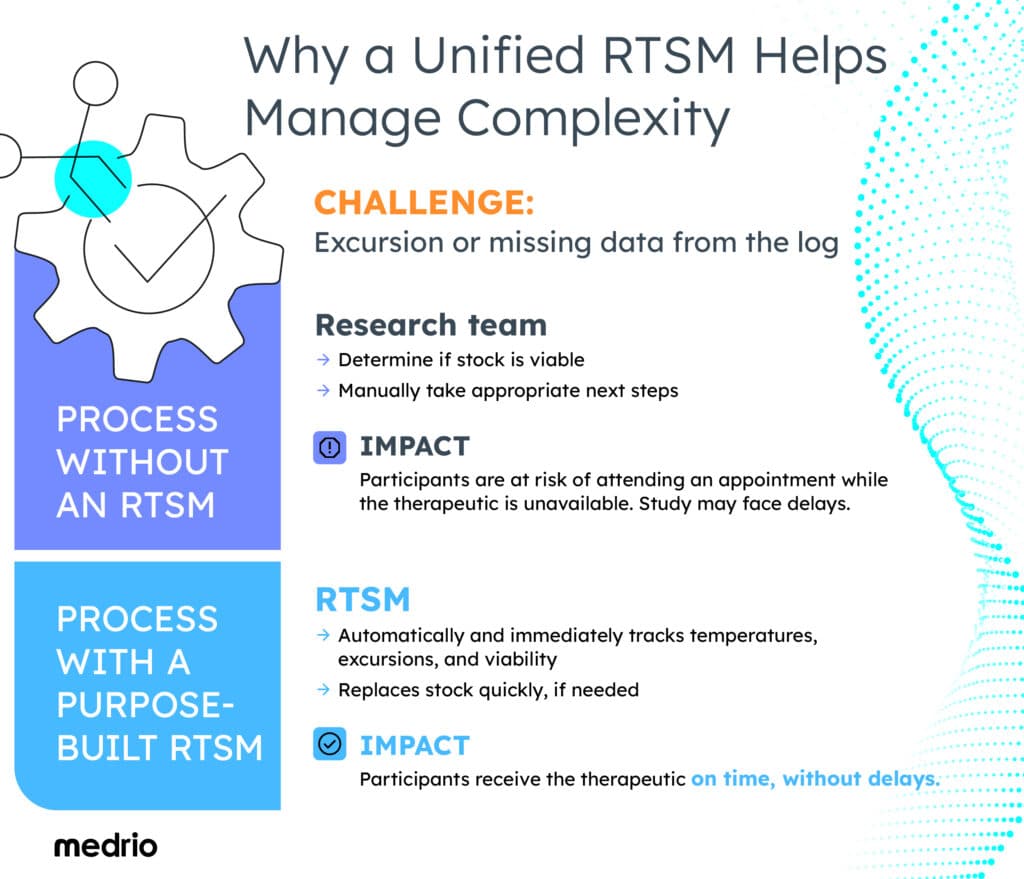
Selecting a Unified Technology Platform to Simplify Processes
When systems are siloed, the entire clinical trial process slows down. For example, manually recording, checking, and interpreting temperature logs can add hours, even days, to study timelines.
In the case of an excursion or missing data from the logs, the sponsor, CRO and investigator must take time to determine whether the stock is still viable. Meanwhile, participants may attend their clinic appointments only to be told that the therapeutic is unavailable, wasting their valuable time. Since many participants live far from clinical sites, placing undue time burdens on them increases dropout rates.
By contrast, a mature, modern RTSM automatically and immediately tracks temperatures, excursions, and viability, ensuring the drugs reach the participants on time. And if drugs do need to be quarantined, an automated RTSM can react immediately, replacing stock quickly and reducing unnecessary delays.
A unified RTSM creates a closed loop in which previously siloed activities are managed simultaneously, from release at the storage and distribution vendor, through shipping and receipt at site, dispensing to the participant, and returns or destruction.
As an automated, cloud-based system, a unified RTSM is faster and less prone to user error than legacy approaches that require manual input, whether paper-based or digital. Modifications take minutes rather than days, and adaptive protocol designs can be managed effectively without long lag times.
How Can You Make Best Use of Staff Resources?
When creativity and flexibility are required, a knowledgeable person can pivot more effectively than a computer program. However, many investigators and site staff are overwhelmed with menial tasks, such as recording and updating data, which can be completed faster and more accurately by an RTSM system.

Creating More Efficient Workflows
Automated systems can free up staff workload and allow them to focus on the irreplaceable aspects of their jobs: human interaction and creative problem-solving.
For example, real-time report generation can be completed through automated, cloud-based RTSM platforms. The program can do the bulk of the work when all goes well. However, if it flags a problem, it can alert the person responsible to investigate it and develop a solution. This streamlined workflow means that the problems find the people rather than the people spending their time looking for problems.
Another example is temperature logs. If every temperature excursion needs to be tracked and evaluated by people, the process can take hours or days. Most logs will report no problems, so staff are just checking for nonexistent issues.
However, a purpose-built RTSM system can automatically record the temperature logs and alert the appropriate CMC or Project Management resources supplying them with a summary of the issue and an accompanying temperature log. This process allows site staff to only check the logs when necessary, rather than spending hours each week poring over them.
Our internal training programs reflect our commitment to maintaining the highest levels of data security. Medrio employees receive thorough training on SOPs for data security and IT-related scenarios, including disaster recovery.
Reducing Staff Workload
Reducing staff workload also helps companies avoid study lags due to high employee-turnover rates.
Staffing shortages and high turnover are endemic to the industry. Wherever companies can decrease staff workload, they can reduce employment costs. Also, automated systems take less time to learn than manual solutions like spreadsheets.
Since site staff frequently train new employees, reducing training times makes those transitions smoother and faster. An RTSM platform that is easy to navigate and learn also allows new staff to onboard quickly.
Are You Optimizing Your Supply Chain?
Ultimately, clinical trial supply chain logistics has a simple goal: get the right medicine to the right participant at the right time. The process itself, however, is far from simple.
From packaging and labeling to shipping, storage, distribution, and, eventually, destruction, the entire life cycle of the IP demands rigorous tracking and tracing. Sponsors and CROs benefit by planning for each of these stages well in advance. When consultants such as Medrio are brought in during the protocol writing stage, they can provide insights that create a more streamlined, cost-effective process that assures compliance.
At Medrio, our services extend beyond data and supply chain management and include protocol review, in which we advise our customers on ways to streamline supply chain logistics through protocol design. As supply chain experts, we can provide feedback, such as altering the packaging plan to allow for greater flexibility or labeling individual products with serial numbers rather than entire batches to improve tracking and tracing.
Clinical trials are increasingly complex, but optimizing your supply chain doesn’t have to be, with the right RTSM partner.
Learn more about how to optimize your supply chain in our guide, “Supply Chain Optimization: Seven Considerations for Your Next Clinical Trial.”