Contributing experts at Medrio: Marc Weinberg, Rare Disease Research Advocate and Senior Director of RTSM
Gene therapy clinical trials represent a pivot point in medicine, offering hope for individuals suffering from genetic disorders and chronic diseases.
By targeting the underlying genetic defects responsible for various ailments, gene therapy promises to treat symptoms and potentially offer cures.
Gene therapy research holds the potential to revolutionize treatment paradigms and improve patient outcomes. Researchers are exploring gene therapies for inherited disorders like cystic fibrosis and muscular dystrophy to complex conditions such as cancer and cardiovascular diseases.
This innovative research requires a reliable technology partner. Explore our clinical trial solutions to learn how Medrio can support your research.
Progress within Gene Therapy
The Human Genome Project is one of history’s most extraordinary scientific feats. By sequencing and mapping the human genome, the project laid the foundation for the current surge of gene therapy advances.
The initial lab experiments in 2021 demonstrated CRISPR’s potential as a gene-editing tool. Just over a decade later, in 2023, a CRISPR-based medicine to treat sickle cell disease and beta-thalassemia was approved. This landmark approval ushered in a new era of genetic medicine.
When the FDA released new policies to advance the development of safe and effective cell and gene therapies, the FDA Commissioner Scott Gottlieb, M.D. stated, “By 2025, we predict that the FDA will be approving 10 to 20 cell and gene therapy products a year based on an assessment of the current pipeline and the clinical success rates of these products.”
Recent Approved Gene Therapies
Recent regulatory approved gene therapies signify a remarkable leap forward in medicine. A new era of hope and possibility is blooming for patients with previously untreatable genetic conditions.
Approvals underscore the culmination of years of rigorous research, innovation, and collaboration between researchers, clinicians, and regulatory agencies.
One of the latest FDA approved gene therapies belongs to Lenmeldy. It is the first-ever treatment for children with metachromatic leukodystrophy, a devastating genetic disorder that is otherwise fatal.
Recent exciting cell and gene therapy approvals include:
- The first gene therapy for adults with severe hemophilia A
- The first cellular therapy for patients with Type 1 diabetes
- The first topical gene therapy for treatment of wounds connected to Dystrophic Epidermolysis Bullosa
- A therapy for patients with blood cancers to use following stem cell transplantation to reduce risk of infection
With each green light from regulatory bodies, gene therapies gain further validation as safe and effective treatments for many genetic disorders.
Each approval brings newfound optimism to patients and their families. New therapeutics inspire confidence in the transformative potential of gene therapy to better the human condition and reshape the future of healthcare.
Learn more in our blog about the modern trial designs and supply chain strategies driving rare disease research.

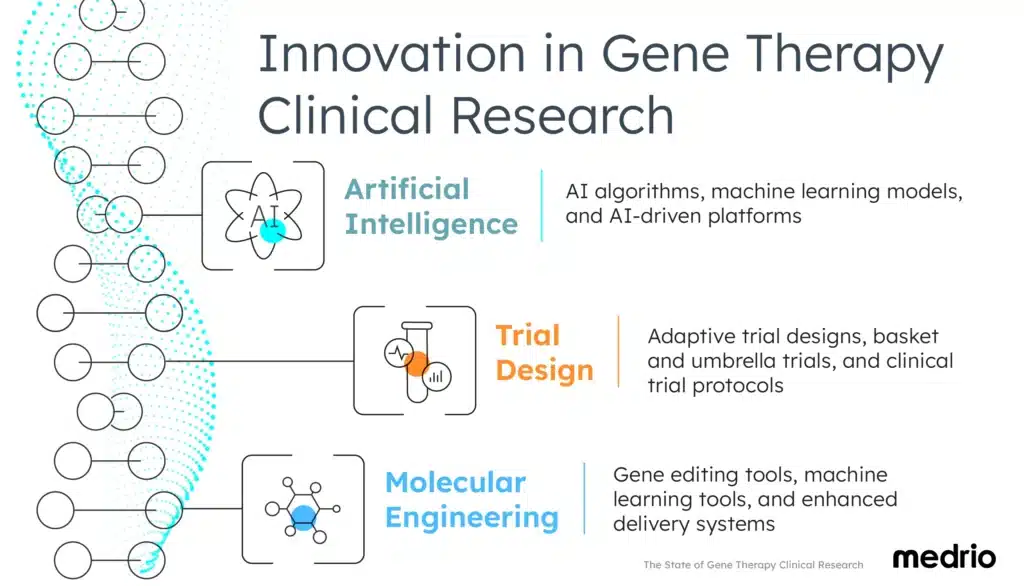
Innovation in Gene Therapy Clinical Trials
The intersection of technology and science is expediting gene therapy clinical research.
Researchers are uncovering solutions to previously unanswered questions. Current advancements in clinical trials are based on artificial intelligence (AI), trial design evolution, and molecular engineering breakthroughs.
Artificial intelligence supports gene therapy innovation
AI breakthroughs have revolutionized the clinical research landscape in gene therapy, promising more precise and efficient treatments.
- AI algorithms have enhanced the identification of potential gene targets. This method expedites the selection of suitable candidates for gene therapy interventions.
- Machine learning models allow researchers to analyze vast genomic datasets and pinpoint genetic variations associated with diseases. This advancement in technology paves the way for personalized therapeutic approaches.
- AI-driven platforms can help predict treatment outcomes. Clinicians can, therefore, tailor interventions to individual patients with greater accuracy and efficacy.
Trial design for gene therapy clinical research
Trial designs in gene therapy clinical research have also evolved significantly, adapting to the complexities of genetic diseases and the unique mechanisms of gene-based treatments.
- Adaptive trial designs, empowered by AI algorithms, allow for real-time adjustments based on accumulating data, optimizing treatment protocols, and minimizing patient risk.
- Basket and umbrella trials offer a multifaceted approach to evaluating gene therapies across different disease subtypes or genetic mutations. These innovative trial types accelerate the pace of discovery, expanding treatment options for patients.
- Clinical trial protocols may possibly be written for individuals rather than groups of participants. This specificity within protocol writing may enable these trials to investigate treatments for an individual patient while still using objective design. This approach results in evidence-based decisions on treatment. Data from multiple ‘N of 1’ trials may be pooled for analysis and results ultimately be applied to a larger group.
Molecular engineering to advance gene therapy
Breakthroughs in molecular engineering are driving exciting breakthroughs in gene therapy.
In tandem with AI advancements, and refined trial designs, the emergence of novel molecules and focus areas has propelled gene therapy research into new frontiers.
- Gene editing tools, like CRISPR-Cas9, offer unprecedented precision in modifying genetic sequences to correct disease-causing mutations.
- Machine learning algorithms are optimizing gene therapy protocols, streamlining the identification of potential targets, and enhancing treatment personalization for patients.
- Enhanced delivery systems are revolutionizing gene therapy by efficiently transporting therapeutic genes to target cells, overcoming previous barriers to treatment efficacy.
Patient Advocacy within Gene Therapy Research
In some cases, patients with genetic disorders and their families become directly involved in shaping clinical research. Some lobby for new treatments. Others advise on how to build effective clinical trials.
For example, there’s the remarkable journey of Pat Furlong. Upon learning of her son’s diagnosis of Duchenne muscular dystrophy (DMD). This devoted mother became an advocate of research for this challenging condition.
Through her non-profit organization, Parent Project Muscular Dystrophy (PPMD), Furlong has spearheaded efforts that garnered hundreds of millions of dollars for research and broader initiatives.
Notably, PPMD played a pivotal role in pioneering gene therapy research and contributed $2.4 million to fund trials, culminating in the FDA approval of the groundbreaking drug Elevidys.
Then there’s the story of Brad Margys. His sons were diagnosed with A-T, a rare genetic disorder that causes severe neurodegeneration. Although his two sons missed the window to receive treatments for A-T, Margys remains dedicated to finding a cure. He is actively involved in fundraising, recruiting researchers, and compiling patient data.
For example, Margys compiled a database containing the sequenced genomes of 235 children with A-T. After sharing it with clinical researchers, they were ultimately able to analyze the patient data, identify genetic mutations causing A-T, and determine treatable mutations.

Partnering in Gene Therapy Clinical Trials for a Better Tomorrow
The current state of clinical research within gene therapy is nothing short of exhilarating.
With groundbreaking advancements in AI-driven approaches, innovative trial designs, and the development of novel molecules, the field is poised for unprecedented growth and impact. Recent regulatory approvals pave the way for transformative treatments, offering hope to millions of individuals affected by genetic diseases worldwide.
As researchers continue to push the boundaries of science and innovation, the future of gene therapy shines brightly. Today’s clinical research within gene therapy promises to revolutionize medicine and improve the lives of countless patients for generations to come.
For this innovative research to be successful, organizations need a reliable technology vendor that supplies flexible gene therapy solutions.