Contributing Experts at Medrio: Ian Davison, RTSM Subject Matter Expert, and Marc Weinberg, Senior Director of RTSM
Randomization and trial supply management (RTSM) is a powerful solution. It can effectively randomize, manage, and control your clinical trial supply chain.
The right RTSM system minimizes risks, while improving efficiencies, regulatory compliance, data visibility, sustainability, patient safety, and more.
As trials grow in complexity, study managers need adaptive, unified RTSM technology to help reduce costs and maintain accurate, real-time data.
Here are the questions this article will answer:
- What is RTSM in clinical trials?
- What should RTSM support?
- Why is RTSM in clinical trials important?
- What should you look for in an RTSM?
- How do you implement RTSM?
- What are compliance considerations for RTSM?
Want to learn more about what to look for in an RTSM partner? Explore our guide on clinical trial supply chain optimization.
What is RTSM in Clinical Trials?
RTSM (Randomization and Trial Supply Management) in clinical trials automates patient randomization and manages the supply chain. It ensures unbiased subject allocation to treatment arms, optimizes inventory management, and streamlines logistics across sites. This technology improves efficiency, reduces waste, and enhances data integrity in clinical research.
The Basics of Randomization in Clinical Trials
Randomization in clinical trials is the process of assigning participants to treatment and control groups. This approach ensures that each individual is assigned to one of the trial’s therapies according to the protocol. The process is unbiased and often blinded.
Randomization allows trial staff to make informed decisions while following protocols. It is the gold standard for evaluating the efficacy of a new therapy and the effectiveness of interventions.
The Basics of Trial Supply Management
Trial supply management is the ability to track inventory. Tracking takes place through packaging, labeling, release, returns, and destruction.
Effective clinical trial supply management ensures the right product is available and allocated to the right participant at the right time. Developing a streamlined supply chain management strategy alleviates staff workload and reduces waste.
5 Ways RTSM Supports Study Managers
Download our helpful infographic showcasing the benefits RTSM gives study managers.
RTSM Technologies and Systems
Technologies to streamline randomizations or manage trial supply have been available since the 1990s. These technologies are known by a variety of names.
They enable tasks including randomization, drug assignment, dose adjustments, and inventory management.

RTSM – Randomization and Trial Supply Management
Randomization and trial supply management (RTSM) is a more recent term to describe a technology system that randomizes, manages, and controls the clinical trial supply chain.
IRT – Interactive Response System
Interactive Response Technology (IRT) is an umbrella term. It refers to both Interactive Voice Response Systems (IVRS) and Interactive Web Response Systems (IWRS).
This technology helps clinical trial sponsors and sites manage participant and drug supply logistics.
IWR or IWRS – Interactive Web Response (System)
IWR is a web-based system providing a user-friendly interface accessible via web browsers.
This system enables real-time updates and tracking of trial activities. A benefit of IWRS is automatic integration with other systems and remote accessibility from anywhere.
IVR or IVRS – Interactive Voice Response (System)
IVRS allowed site staff to randomize patients via voice-based systems, such as phones.
Before internet connections were commonplace, site staff could interact with a database via IVRS by pressing keypad buttons on a phone. IVRS evolved to dispense investigational product (IP) and resupply at clinical trial sites.
IXR or IXRS – Interactive Voice/Web Response System
IXRS combines IVR technology with web-based interfaces.
It provides multiple communication and data entry channels for trial participants with limited internet access. Participants can interact with IXRS through phone calls or web portals to report data or receive instructions.
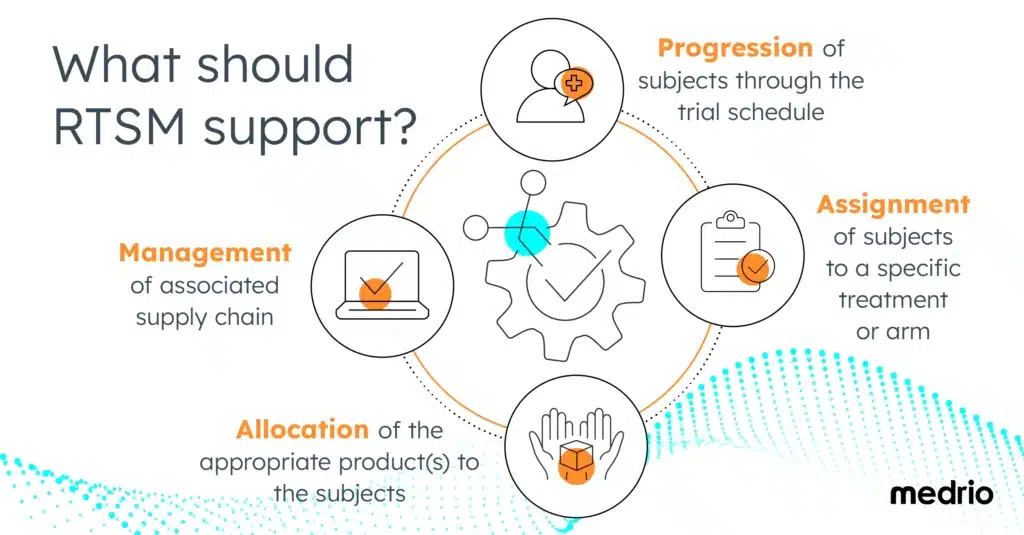
What Should an RTSM Support?
An RTSM should provide features that help sponsors, CROs, and research sites to manage and randomize participants while managing the supply chain.
RSTM should support the following:
- Progression of subjects through the trial schedule
- Assignment of subjects to a specific treatment or arm
- Allocation of the appropriate product(s) to the subjects
- Management of associated supply chain
Participant management
RTSM streamlines the participant’s journey through a trial.
The technology delivers participant-specific dosing instructions. A robust RTSM allows for easy participant eligibility confirmation, balanced cohort maintenance, and dosing instruction and schedule distribution.
The right RTSM can:
- Automate dosing and allocating instructions for site staff
- Capture basic visit information, demographic data, and inclusion/exclusion criteria
- Manage cohorts
Randomization
The right RTSM is capable of supporting a wide range of randomization types and complexities.
Replacing manual randomization methods with technology guarantees bias-free study data. RTSM also automates the process of maintaining balanced cohorts, even if participants drop out.
The right RTSM can:
- Automate randomization assignments
- Send confirmatory emails and reports as randomization progresses
- Implement a broad range of randomization types
Supply chain management
RTSM tracks inventory from release at the depot through delivery to sites and subjects, and eventual return and destruction.
Study teams have complete visibility into the product journey, allowing them to efficiently view and manage inventory levels.
The right RTSM can:
- Alert for shipments, depots, site inventory, subject visit, late dispatch, and receipt of shipment warning
- Automate expiry date management, low shipment notifications, and site inventory resupply orders
- Manage temperature controls, including enforced temperature log uploads and excursion notifications
Why is RTSM in Clinical Trials Important?
Randomization and trial supply management encompasses several basic functions. Although some functions can be done manually, automation by an electronic solution improves trial efficiency and accuracy.
RTSM is important in trials because it:
- Provides comprehensive randomization and trial supply management capabilities
- Ensures accurate, unbiased allocation of treatment assignments
- Offers efficient inventory control
What Are the Benefits of an RTSM?
Beyond randomization, the right RTSM gets the investigational product (IP) on shelves when and where it’s needed.
The main benefits you can expect from an RTSM are:
- Improved control through a centralized system
- Increased real-time visibility
- Less waste and supply excess
- More streamlined supply approvals
- Better protocol compliance and fewer violations
- Access to configurable, real-time reports
- Ability to dissociate product labeling from subjects
- Option for adaptive study design

How to Choose the Right RTSM Software
It can be hard to know where to begin when choosing the right RTSM system for your studies.
The process for selecting an RTSM vendor may include:
- Collating the Protocol, Clinical Trial Supplies Plan, and the Pharmacy Manual.
- Asking the RTSM vendor to review these documents for a consultation.
- Requesting a demo from the RTSM vendor.
- Determining system resiliency.
- Checking if the software can interface with any other relevant stakeholders in the trial, particularly the logistics and EDC vendors.
- Confirming the vendor offers fast, reliable and competent support.
- Reviewing quality assurance.
- Reviewing timelines and resources.
Questions to ask when reviewing a vendor:
- Can the vendor add value to your trial, optimize site workflow, supply usage and process visibility?
- Is the workflow clear and intuitive?
- Does the system have the functionality you need?
- Has the vendor demonstrated the domain expertise and problem-solving skills to deliver the right solution?
- How does the system deal with incidents, user errors, protocol amendments, and any particular risks associated with your project?
- Does the vendor have the appropriate procedures and validation records to comply with global and regional regulatory requirements?
- Can the vendor deliver to the required timelines and within the available budget?
For more in-depth questions to ask your team and a potential RTSM vendor, explore our RTSM readiness guide.
What to Look for in an RTSM
In a dynamic and complex trial environment, the right RTSM tool manages supply chain issues before they become problems.
The most important criteria for selecting an RTSM vendor are:
- Integration with EDC, ePRO, CTMS, and other vital systems
- Start-up timelines, speed of build
- A collaborative approach with early engagement
- Ease-of-use for clinician team
- Ease-of-use for site team
- Configurability for study setup
Advanced features enable you to:
- Configure allocation rules
- Automate shipment approvals
- Receive issue notifications to streamline the supply chain process
There can also be specific considerations for different therapeutic areas. Click the links to learn more about supply chain solutions for oncology trials and supply chain solutions for rare disease trials.

How to Implement RTSM in Your Clinical Trial
Getting the most out of an RTSM system requires that the software is aligned with your master protocol and your data management processes.
Planning and design considerations for RTSM
Your RTSM sits at the intersection of your master protocol and data management processes.
Key considerations to review with your RTSM partner before implementation include:
- Trial basics
- Participant management
- Dosing and dispensing
- Cohort management
- Schema and treatment plan
- IP packaging and management
- Data management and compliance
Setting Up an RTSM System: Simple Step-by-Step Guide
Your RTSM vendor should act as a partner and guide you during your RTSM system set up.
Key implementation guidance areas from your RTSM partner should include:
- Determining the scope of the system, ensuring that your protocol, supplies plan, and pharmacy manual are supported and aligned
- Identifying a supply chain optimization strategy to make the best use of your resources
- Partitioning RTSM workflow from EDC data, and ensuring the right data and process lives in the right system
- Delivering a validated system, ensuring the appropriate SDLC is followed with full visibility
- Ensuring protocol amendments are dealt with efficiently
RTSM Best Practices and Compliance
Although RTSM data won’t be shared with regulators, it can influence shared data.
RTSM systems don’t capture source data. However, they do interact with a primary data source, such as an electronic data capture (EDC) solution.
Integrating RTSM with other systems can remove risks from manual, error-prone processes. They can also create greater real-time transparency of supply status.
Compliance best practices for RTSM systems include:
- Ensuring all data systems interact seamlessly and securely
- Selecting a vendor with supply chain experts
- Integrating the RTSM with other compliant system
The Future of RTSM in Clinical Trials
RTSM has existed in some form or another for the last 30 years. As this technology matures, RTSM clinical trials will evolve to meet the ever changing needs of clinical research.
There are emerging trends in randomization and supply management as the technology is becoming more sophisticated.
Sponsors, CROs, and sites can expect to see RTSM advances including:
- Greater use of cloud-based solutions for ease-of-use and scalability
- An increased focus on patient safety
- More collaboration across stakeholders

RTSM FAQs
What does RTSM stand for?
RTSM stands for randomization and trial supply management.
What is clinical trial supply management?
Clinical trial supply management involves planning, forecasting, procurement, distribution, and destruction.
What is RTSM in clinical trials?
Randomization and trial supply management (RTSM) is a tool that can effectively randomize, manage, and control your trial supplies.
What kinds of trials benefit most from an RTSM?
Studies of all sizes and phases can benefit. Advanced features help you quickly align to your sites’ storage capabilities and reduce waste using algorithms that automate supply management. You can remove bias; enhance oversight of incidents, accountability, returns, and destruction; and eliminate duplicate data entry and reconciliation.
Advance Your Clinical Trials with Medrio
In a dynamic and complex trial environment, the right RTSM solution supports participant management, randomization, and supply chain management.
Medrio provides an integrated suite of solutions on a unified platform, including RTSM, CDMS/EDC, eCOA/ePRO, and eConsent. This unified platform facilitates comprehensive study oversight and live data synchronization, presented in a modern, user-friendly interface.
Explore our clinical trial supply chain optimization guide to learn from our experts about how to reduce IP waste, build supply chain strategies, and select a robust RTSM partner.