Streamline all aspects of the informed consent process and enable participants to consent from anywhere — remotely or onsite.
Capture paper or electronic informed consent forms (ICFs) simultaneously.
EU-compatible and 21 CFR Part 11-compliant solution that meets ALCOA++ standards.
Enhance participant comprehension and retention with images, video content, quizzes, and FAQ sections.
Automate participant consent requests and site document assignments, ultimately reducing site burden.
Our flexible, web-based solution supports electronic and paper-based processes, allowing you to prioritize the patients’ experience and the sites’ needs. Efficiently deliver consent forms; capture electronic signatures; and implement in-clinic, remote, or hybrid workflows.
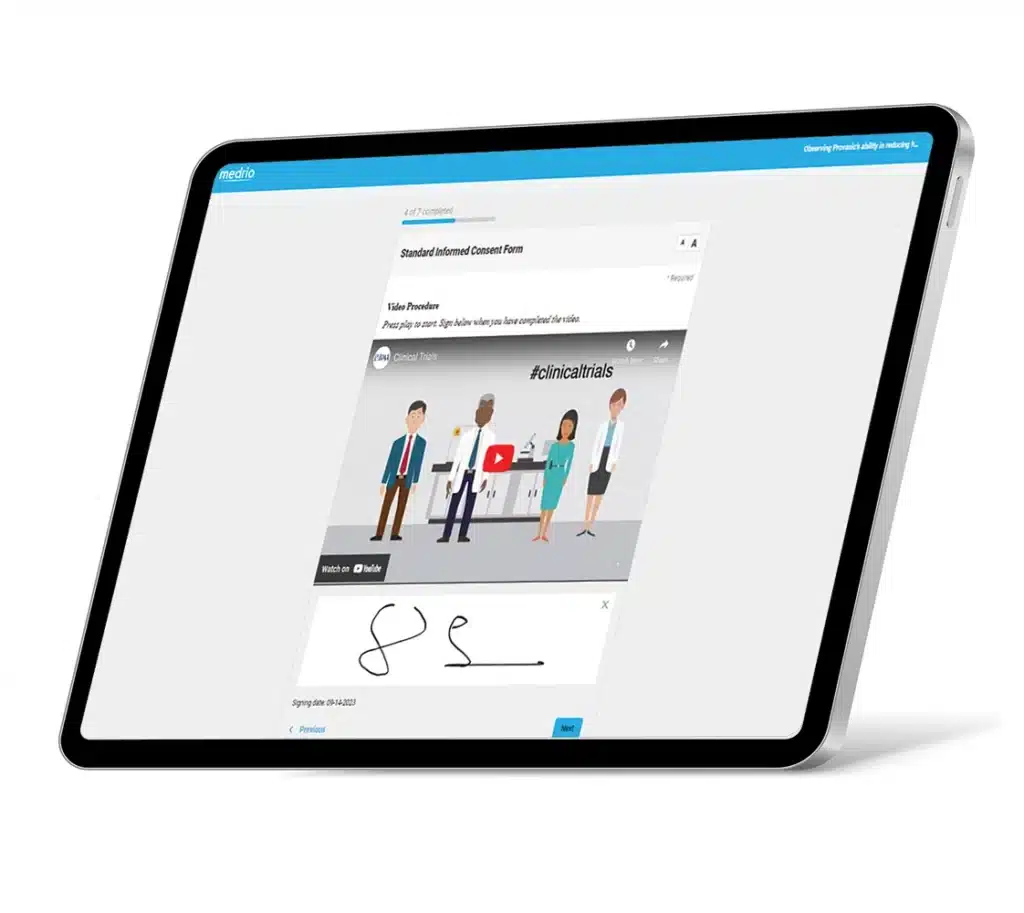
Built-in flexibility means minimal workflow disruptions for sites and more autonomy for participants. You can upload paper consent, download and print unsigned consent forms, and collect eConsent across geographies with different regulations, making it easy to customize processes.
Find out why Medrio has a 98% customer retention rate.
“One of the easiest systems to implement mid-study updates. Queries are easy to find and respond to. Audit trails are straightforward as well.”
Dan Pontoriero
“They are reliable and meet timelines. I got the same team members from beginning to end. Working with Ramana was a breath of fresh air!”
Claudia Ramos
“Medrio provides a system that is versatile and simplifies the complexities of data collection and management for administrators and data managers.”
Anonymous






Sign up to have our the latest insights delivered to your inbox.