Solutions > eTMF
Electronic Trial Master File (eTMF) Software
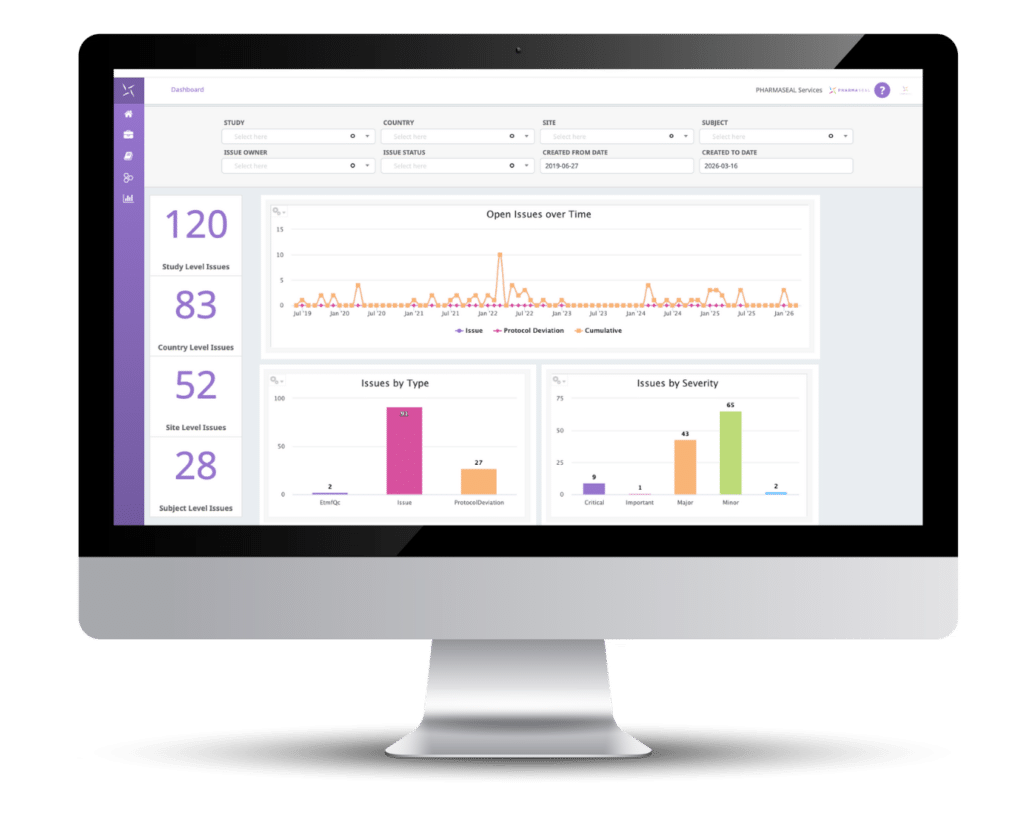
Your electronic trial master file (eTMF) should do more than store documents. Medrio’s eTMF software, powered by Engility®, is the compliance engine that automatically files documents, tracks completeness, and keeps your study inspection-ready from Day 1.
Continuous Readiness
Maintain inspection readiness from day one. Track missing documents and expiry dates automatically.
Unified Workflow
Our eTMF is unified with Medrio CTMS to share and reduce data duplication and automate document workflows.
Auditor-Friendly View
Provide selective access to external auditors and streamline audits and inspections to meet your regulatory obligations.
Automated Regulatory Compliance


Seamless CTMS & EDC Integration
Collaborative Site Exchange
What Medrio Customers Are Saying
Find out why Medrio has a 98% customer retention rate.
“Built with the builder in mind.”
“One of the easiest systems to implement mid-study updates. Queries are easy to find and respond to. Audit trails are straightforward as well.”
Dan Pontoriero
“The entire team is exceptional.”
“They are reliable and meet timelines. I got the same team members from beginning to end. Working with Ramana was a breath of fresh air!”
Claudia Ramos
SR. DIRECTOR, CLINICAL OPERATIONS
“Data simplified!”
“Medrio provides a system that is versatile and simplifies the complexities of data collection and management for administrators and data managers.”
Anonymous
OUR INTEGRATED SUITE OF SOLUTIONS
Validate faster with the industry’s most trusted, no-code EDC. Get enterprise-grade data rigor without the complexity or cost.
Proudly Serving Life Science Innovators
Our commitment extends beyond providing solutions; it’s about being a part of a collective effort to drive positive change and innovation in critical sectors of the life science industry.
Frequently Asked Questions
How do I evaluate & choose the best eTMF software vendor?
The best eTMF systems in 2026 have evolved from digital filing cabinets into active oversight tools. When evaluating eTMF software vendors, mid-sized biotechs should weigh the “enterprise burden” of legacy providers against Medrio’s focus on agile efficiency. Look for a partner that prioritizes speed and control, allowing for a faster go-live and intuitive document management without the heavy manual service models or rigid configurations of older systems.
What is the difference between a Trial Master File (TMF) and an Electronic Investigator Site File (eISF)?
An eISF is used for site-specific documentation. The TMF is used by the Sponsor (or CRO on behalf of the Sponsor) to store all of the essential trial files. This includes some documents that are also stored in an ISF.
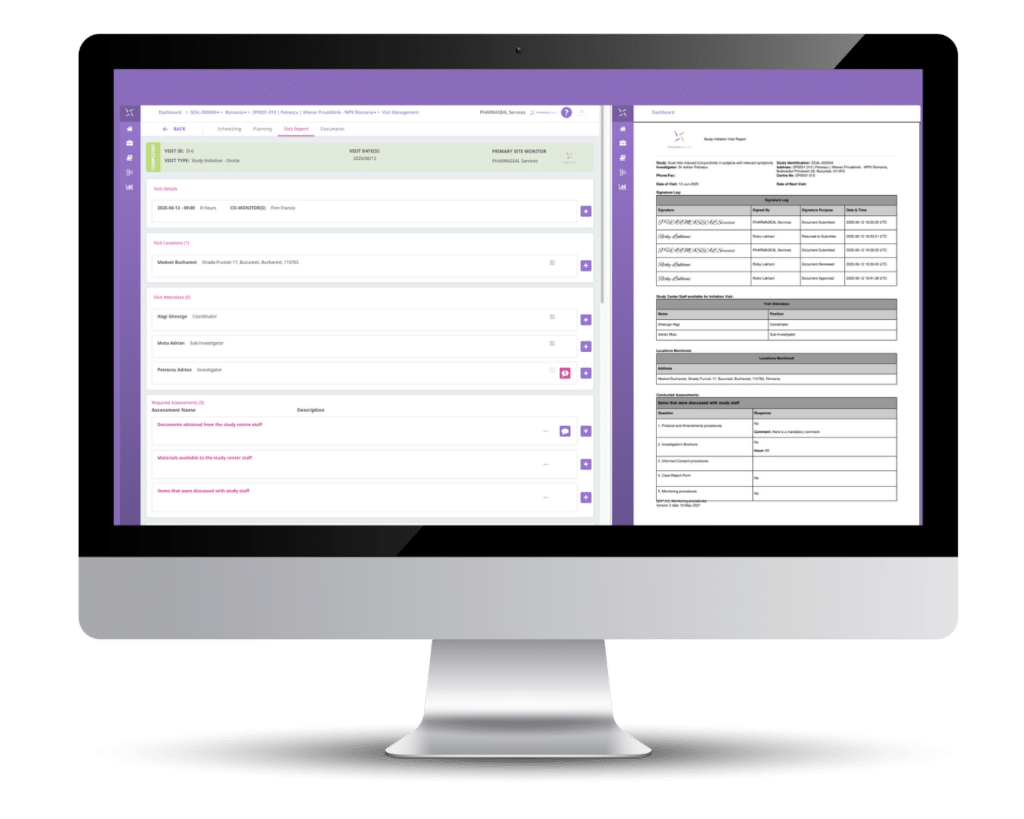
Medrio’s eTMF does not have any ISF capabilities. However, with the appropriate permissions, sites can upload study pertinent documents to the eTMF, usually in a centralized folder that restricts who can see or access what documents within that and other locations.
Why is Medrio among the best eTMF vendors in 2026?
While legacy giants like Veeva Vault eTMF offer robust features, they often come with high overhead and complexity. Medrio is the right-sized eTMF partner for organizations seeking agile, modern solutions with streamlined workflows. We provide the same regulatory depth as enterprise systems but with an intuitive user experience. This allows clinical ops leads to manage the system themselves with confidence.
How does Medrio eTMF integrate with Medrio CTMS?
Modern clinical buyers prioritize top systems integrating eTMF and regulatory data workflows to ensure a unified flow. Medrio’s eTMF and CTMS share global setup information, such as contract information and site information. This allows for faster setup time in both modules.
Is Medrio an effective eTMF solution for smaller biotechs & CROs?
Yes. Medrio offers affordable eTMF solutions by eliminating unnecessary features and costs associated with larger competitors. Instead, our model features transparent, scalable eTMF pricing that fits your study’s budget constraints.
Does Medrio eTMF ensure compliance with ALCOA-C and 21 CFR Part 11 / Annex 11?
Foundational eTMF compliance is a non-negotiable requirement for an FDA audit of clinical trials. Medrio goes beyond industry best practice to strictly enforce ALCOA-C clinical research principles. To meet FDA 21 CFR Part 11 and EU Annex 11 standards, our system provides an audit trail to ensure total data integrity during global regulatory inspections.
How does Medrio eTMF support inspection readiness?
Medrio eTMF helps you track the health and completeness of your clinical trial. Host an FDA or EMA auditor at a moment’s notice with total confidence in your document completion.
What is the timeline for eTMF implementation?
The initial setup of Medrio eTMF takes approximately a week. Setup for subsequent studies can be completed in a matter of days.
Upholding the Highest Compliance Standards




