Solutions
Our Solutions
A Suite of Clinical Trial Solutions for Every Study
Empowering life science innovators with dynamic technology and high-touch clinical research support.
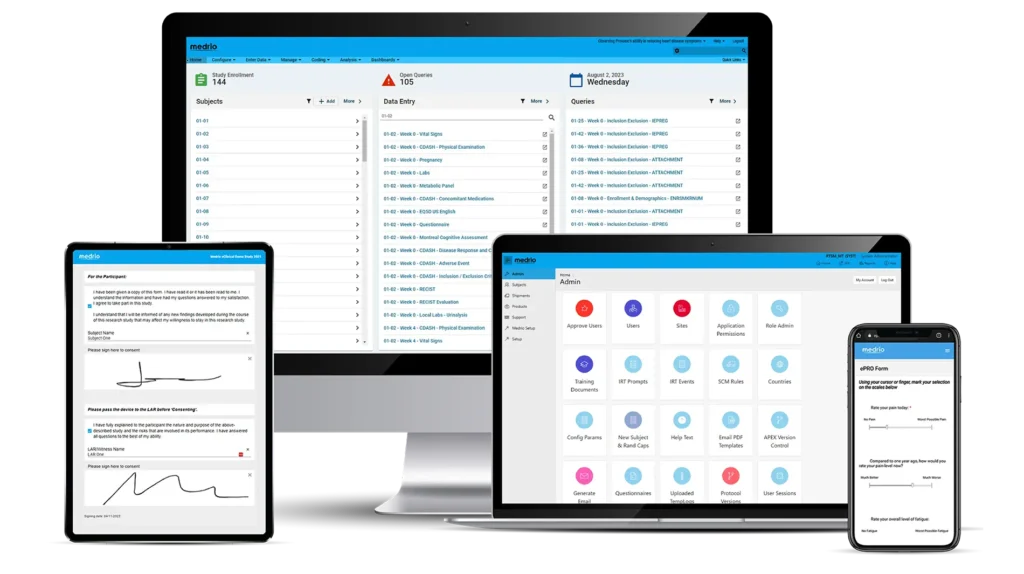
Our Offerings
Validate faster with the industry’s most trusted, no-code EDC. Get enterprise-grade data rigor without the complexity or cost.
Say goodbye to overly complex solutions that are slow to implement, hard to learn, and unable to adapt. Discover the difference of a fully integrated platform.
Control
Maintain autonomy and control of your study from end to end.
Quality
Advanced features for improved data quality and regulatory readiness.
Speed
Designed for the ease of use and efficiency you need.

Enhance the Trial Participant Experience
Achieve faster enrollment, higher retention, and continuous participant engagement, all with real-time study oversight.
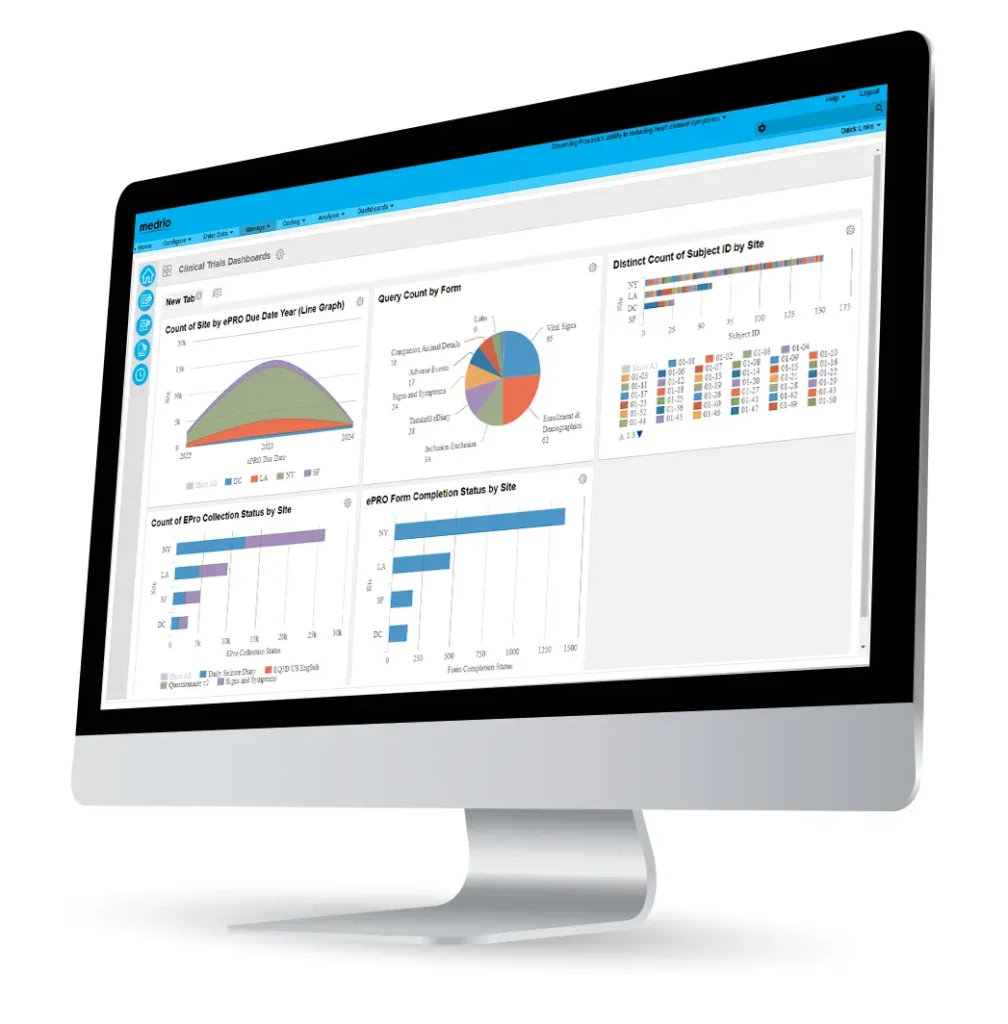
Control Your Clinical Research Data
Experience end-to-end autonomy with advanced controls, real-time data synchronization, and dynamic reporting features.
Accelerate the Time to Market
Implement flexible protocol designs, build eCRFs in minutes, automate randomization, and more. Move faster with solutions that work with you.
What Medrio Customers Are Saying
We offer 24/7 customer support and we have a 98% customer satisfaction rating.
“Built with the builder in mind.”
“One of the easiest systems to implement mid-study updates. Queries are easy to find and respond to. Audit trails are straightforward as well.”
Dan Pontoriero
“The entire team is exceptional.”
“They are reliable and meet timelines. I got the same team members from beginning to end. Working with Ramana was a breath of fresh air!”
Claudia Ramos
SR. DIRECTOR, CLINICAL OPERATIONS
“Data simplified!”
“Medrio provides a system that is versatile and simplifies the complexities of data collection and management for administrators and data managers.”
Anonymous
Proudly Serving Life Science Innovators
Our commitment extends beyond providing solutions; it’s about being a part of a collective effort to drive positive change and innovation in critical sectors of the life science industry.
Upholding the Highest Compliance Standards




