A Partnership Program Built for CRO Growth
Designed to extend CRO capacity without adding internal overhead

A LOOK AT SOME OF OUR PARTNERS
Why join?
Scale and grow without stretching lean teams too thin. This program adds dedicated commercial and delivery support, enabling CROs to take on more work while staying focused on execution.
Enablement training to facilitate successful setup, study build standards and libraries
Co-marketing opportunities and sales support with ongoing education and resources.
Transparent, tiered pricing with rewards for growth.
Change management support to help teams integrate Medrio efficiently
What Our CRO Partners Say
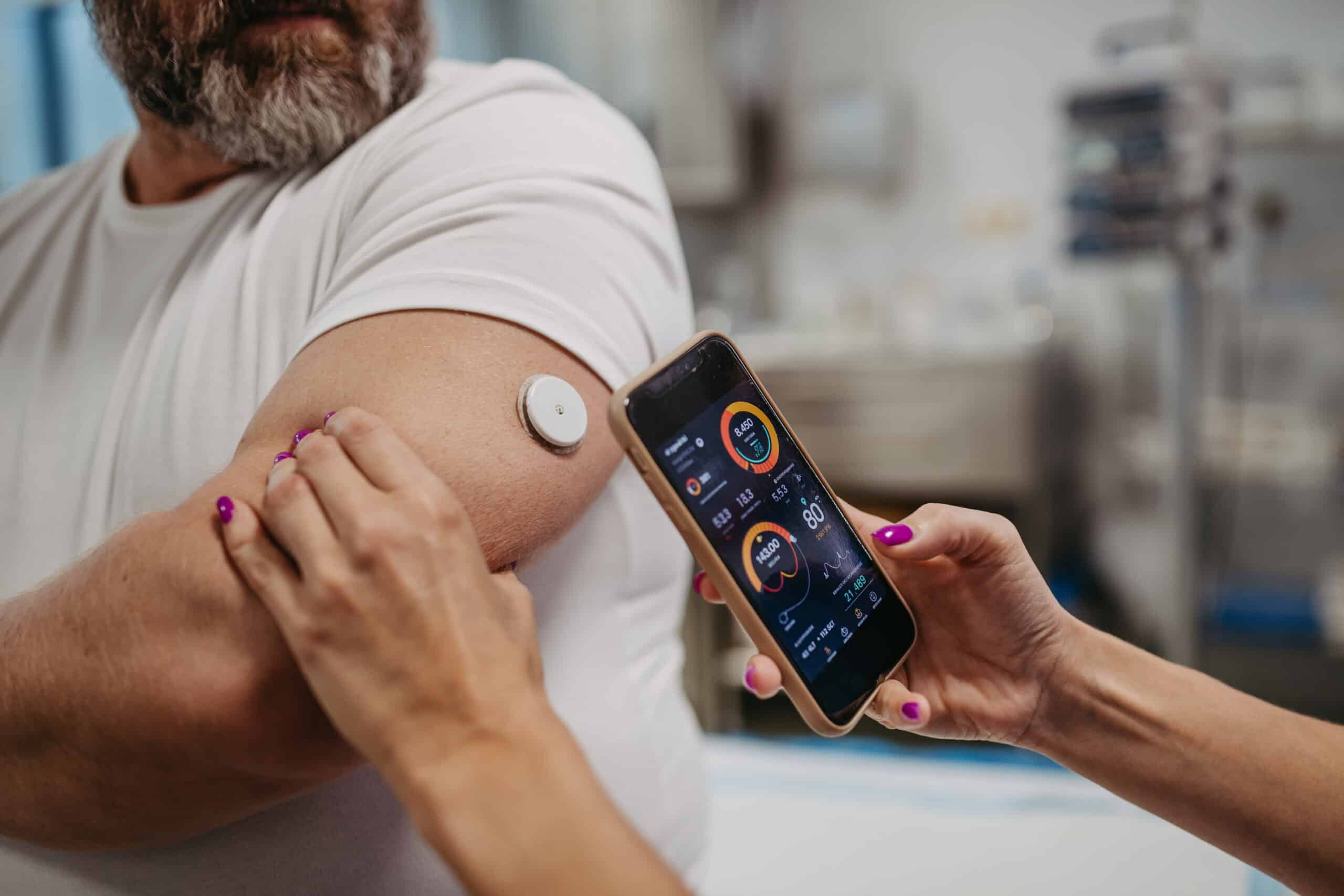
Our relationship with Medrio helps us decrease timelines, especially with regard to study builds. Plus, real-time data management keeps our data clean and decreases database lock time.
Director of Clinical Operations, CTRG
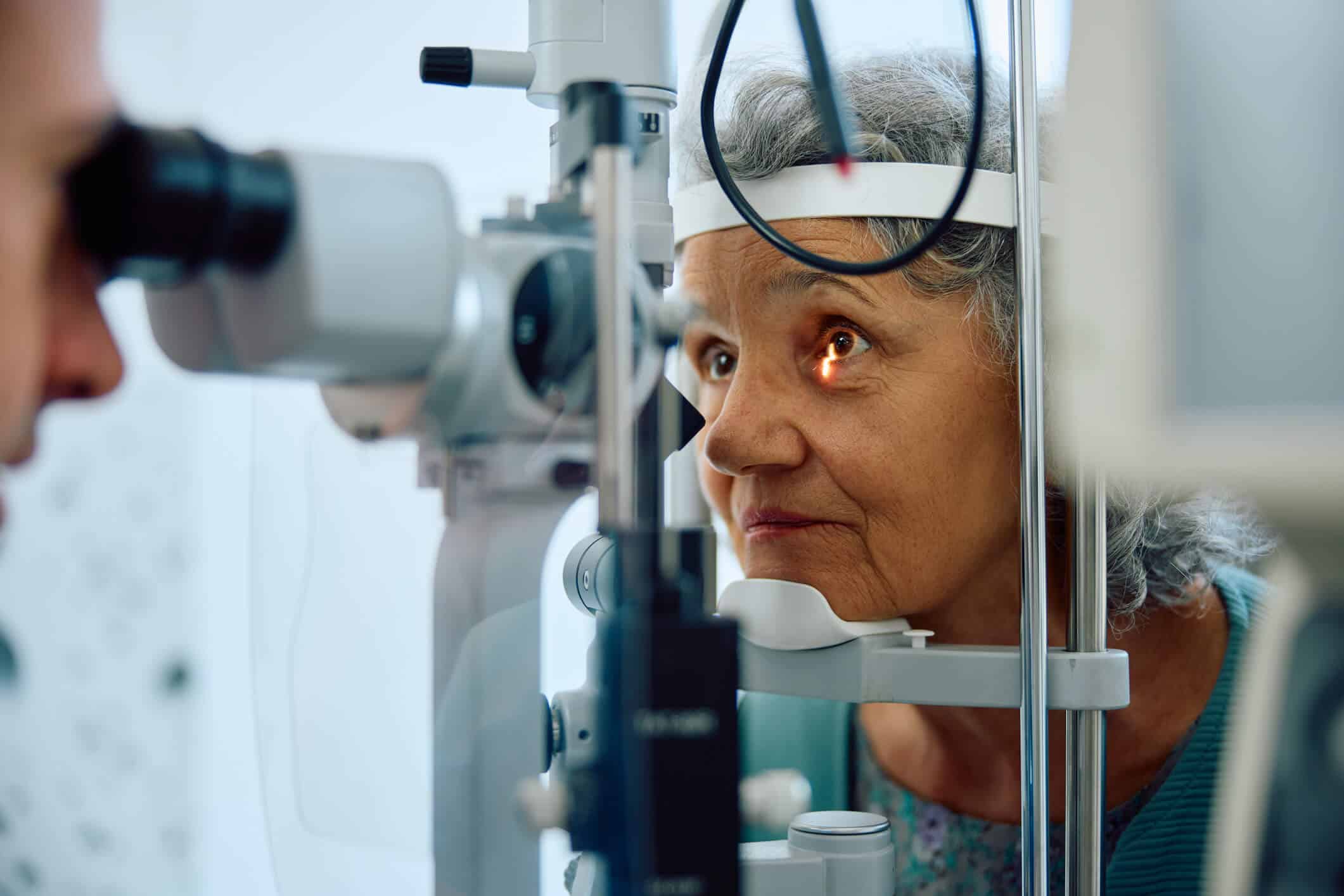
The team was readily available for review calls. Leading up to go-live, we needed ad hoc support, and the team was very available to support us. We really leaned on their expertise.
Senior Clinical Data Manager, CliniLabs


Real-time visibility is key—no waiting, no frustrations. Immediate access to data ensures timely decision-making, preventing delays in critical activities such as safety review committee meetings.
Director of Clinical Data Management & Biometrics, Vanguard Clinical

Medrio sets itself apart through its exemplary customer care and support, establishing a genuine working partnership. There is always someone available, assuring us that we are not alone.
Head of Data Management, MAC Clinical Research
Medrio’s validation environment has been invaluable. We can test changes in a mirrored live environment, anticipate the impact on live data, and ensure seamless transitions.
Senior Technical Clinical Data Manager, DP Clinical

Our relationship with Medrio helps us decrease timelines, especially with regard to study builds. Plus, real-time data management keeps our data clean and decreases database lock time.
Director of Clinical Operations, CTRG
The team was readily available for review calls. Leading up to go-live, we needed ad hoc support, and the team was very available to support us. We really leaned on their expertise.
Senior Clinical Data Manager, CliniLabs

Real-time visibility is key—no waiting, no frustrations. Immediate access to data ensures timely decision-making, preventing delays in critical activities such as safety review committee meetings.
Director of Clinical Data Management & Biometrics, Vanguard Clinical
Medrio sets itself apart through its exemplary customer care and support, establishing a genuine working partnership. There is always someone available, assuring us that we are not alone.
Head of Data Management, MAC Clinical Research
Medrio’s validation environment has been invaluable. We can test changes in a mirrored live environment, anticipate the impact on live data, and ensure seamless transitions.
Senior Technical Clinical Data Manager, DP Clinical





