Solutions > CDMS/EDC
Electronic Data Capture (EDC) Software
More than just an electronic data collection tool for clinical trials. Our EDC software combines an intuitive user interface, seamless online and offline data capture capabilities, and comprehensive data management in a single solution.
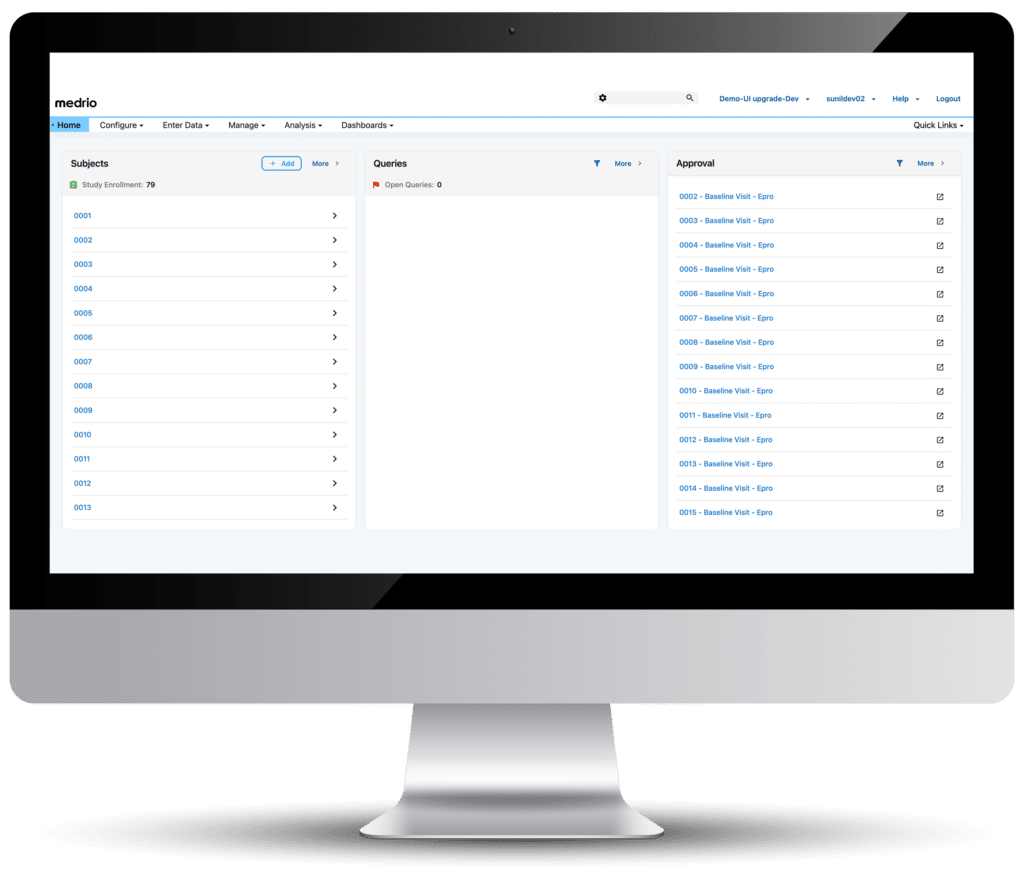
Real-Time Clinical Trial Data Access
Access data effortlessly with dynamic reports, easy data exports, and quick eCRF creation capabilities.
Mid-Study Changes with Zero Downtime
Test changes thoroughly before implementation to ensure seamless integration without downtime.
Easy To Learn, Fast To Deploy
Go live in hours to days with our code-free base and study copy functionality, ensuring a quick study setup.
Accelerate Your Clinical Trial
Build Studies in Weeks, Not Months
When your data capture strategy is built on the right technology foundation, anything is possible — including shorter timelines that allow you to scale with ease.
Secure The Path to Clinical Trial Approval
In clinical research, the data you collect is as pivotal as the therapy asset itself. Medrio CDMS/EDC is purpose-built to safeguard your clinical data with advanced quality, security, and compliance controls.


Turn Time Saved Into Market Advantage
What Medrio Customers Are Saying
Find out why Medrio has a 98% customer retention rate.
“Built with the builder in mind.”
“One of the easiest systems to implement mid-study updates. Queries are easy to find and respond to. Audit trails are straightforward as well.”
Dan Pontoriero
“The entire team is exceptional.”
“They are reliable and meet timelines. I got the same team members from beginning to end. Working with Ramana was a breath of fresh air!”
Claudia Ramos
SR. DIRECTOR, CLINICAL OPERATIONS
“Data simplified!”
“Medrio provides a system that is versatile and simplifies the complexities of data collection and management for administrators and data managers.”
Anonymous
OUR INTEGRATED SUITE OF SOLUTIONS
Validate faster with the industry’s most trusted, no-code EDC. Get enterprise-grade data rigor without the complexity or cost.
Scale from Phase I to post-market. Improve patient retention with flexible, web-based data collection that connects directly to your EDC.
Proudly Serving Life Science Innovators
Our commitment extends beyond providing solutions; it’s about being a part of a collective effort to drive positive change and innovation in critical sectors of the life science industry.
Frequently Asked Questions
What is an Electronic Data Capture (EDC) system, and how does it relate to CDMS in clinical trials?
An Electronic Data Capture (EDC) system is used to collect, manage, and clean clinical trial data electronically. An EDC is also capable of integrating with remote data capture technologies that support decentralized clinical trials. Traditionally, EDC was one component of a broader Clinical Data Management System (CDMS).
Medrio CDMS/EDC functions as a modern, unified platform, combining EDC functionality with core CDMS capabilities such as data validation, query management, audit trails, and database lock. All these benefits occur without the complexity of legacy CDMS tools.
What makes Medrio CDMS/EDC the best clinical data management platform for small to mid-size sponsors and CROs running modern trials?
Medrio CDMS/EDC is designed specifically for today’s clinical trials, with a cloud-based, intuitive interface that reduces build time and operational burden.
Unlike CDMS platforms that rely on custom coding, Medrio CDMS/EDC enables rapid study configuration, flexible mid-study changes, and streamlined workflows through point-and-click configuration. This helps teams move faster while maintaining compliance and data quality/security.
How does Medrio CDMS/EDC ensure compliance, data security, and inspection readiness for clinical trials?
Medrio CDMS/EDC is designed to support regulatory compliance and data quality throughout the clinical trial lifecycle. The platform includes validated electronic records and signatures, full audit trails, role-based access controls, and secure data encryption to align with FDA 21 CFR Part 11, ICH E6(R2), HIPAA, GDPR, and other global requirements.
Medrio CDMS/EDC supports electronic data capture at the point of entry in ways that align with modern eSource expectations, helping reduce reliance on paper documents while maintaining full traceability. Robust data backup, disaster recovery, secure record access, and flexible data exports help ensure inspection readiness and long-term data accessibility.
What types of organizations and trials does Medrio CDMS/EDC support?
Medrio CDMS/EDC supports clinical trials across MedTech, Biotechnology, Pharmaceutical, CRO/Consulting, and Animal Health organizations, from early feasibility studies through pivotal and post-marketing trials.
The CDMS/EDC platform is well suited for small and mid-sized teams as well as growing organizations that need enterprise-grade data management without enterprise-level complexity.
How does Medrio CDMS/EDC compare to platforms like Medidata Rave, Veeva Vault, Castor, OpenClinica, and REDCap?
Compared to traditional enterprise CDMS platforms, Medrio CDMS/EDC emphasizes speed, usability, and flexibility. Teams often choose Medrio for faster study startup, easier eCRF design, and more responsive mid-study updates.
Compared to free, low-cost, academic, or open-source tools, Medrio CDMS/EDC provides stronger validation, scalability, and regulatory support required for regulated clinical trials.
Does Medrio CDMS/EDC support fast study startup and mid-study changes?
Yes. Medrio CDMS/EDC is known for rapid study builds and efficient mid-study amendments. Study teams can implement form updates, logic changes, and protocol-driven modifications without lengthy revalidation cycles, reducing downtime and keeping studies on track. Customers have full control over these processes, without relying on Medrio as a vendor.
How does Medrio CDMS/EDC streamline eCRF design, validation, and database lock?
With Medrio CDMS/EDC, fast study start-up timelines are supported by no-code, point-and-click eCRF builds that don’t require specialized resources to configure and launch.
Medrio CDMS/EDC supports continuous data review and validation throughout the study, helping teams reduce late-stage cleanup at milestones such as Last Patient Last Visit (LPLV). Built-in edit checks, real-time validation, and efficient query workflows allow issues to be identified and resolved earlier.
Flexible mid-study updates and rolling database locks help teams avoid unnecessary rework, enabling faster progression from LPLV to database lock and shorter timelines for analysis and reporting.
Can Medrio CDMS/EDC support adaptive, complex, hybrid, or decentralized clinical trial (DCT) designs?
Medrio CDMS/EDC is flexible enough to support adaptive protocols, complex visit schedules, hybrid models, and decentralized trial components. Its configurable workflows and integration capabilities allow sponsors to tailor data collection to evolving study designs and operational needs.
Does Medrio CDMS/EDC support integrations with other clinical systems and standards?
Does Medrio provide training and support for CDMS/EDC users?
Yes. Medrio provides onboarding, enablement, and ongoing support for customers using Medrio CDMS/EDC. Customers receive study setup guidance, best practices for data management workflows, and responsive assistance throughout the study lifecycle to help teams use the platform effectively.
Learn more at the Medrio Community forum.
Upholding the Highest Compliance Standards




