Long before COVID-19, recruiting and retaining patients for clinical trials was a growing problem with 85% of all trials failing to recruit enough patients and 80% delayed due to recruitment problems and high drop-out rates. To combat this growing issue, the industry is shifting its focus to a patient-centric approach that incorporates the needs of patients into clinical trial designs, making it easier for them to participate.
But is the industry ready to take the leap and fully embrace the final frontier of patient-centricity: decentralized trials?
Patient-facing technologies are playing an increasingly important role in the future of virtual and hybrid trials—especially for new drugs and medical devices. They are enabling remote patients to participate in clinical trials where they haven’t been able to before. Despite some initial resistance, the pandemic has resulted in more sponsors and CROs adopting decentralized approaches.
By removing the necessity for brick-and-mortar sites, sponsors are opening up the possibility for patients to engage online, at home, or even through local labs and health care sites. In short, decentralized clinical trials are leveraging novel technologies to create options for trial participants outside of the conventional clinical setting by improving access and convenience.
They’re doing this through a combination of solutions including electronic consent forms, remote patient monitoring and diagnostics, direct data capture, and direct-to-patient drug distribution. These remote technologies are helping clinical researchers remove heavy burdens for their patients, while also overcoming the biggest challenge of clinical trials: patient recruitment and retention. We will explore how decentralized studies leverage patient-centric tools to ultimately strengthen their trial participation pool.
The Benefits of a Patient-Centric Approach
Patient recruitment and retention continue to be the longest and most arduous part of the clinical trials process. One leading reason is that the industry has not kept up with the way their patients use and interact with technology.
A study titled “The Future of Drug Development” by the Economist Intelligence Unit1 found trials that adopted patient-centric technology were more efficient at recruiting trial participants. In fact, the study which examined nearly 4,000 phase II and III trials, found that patient-centric trials took an average of four months to recruit 100 patients, versus seven months for traditional trials.
On top of that, the study found that drugs developed with patient-centric, decentralized technology were 19% more likely to go to market than traditional in-clinic methods. The numbers were even more staggering for oncology trials, which were 32% more successful using patient-facing, adaptive technology.
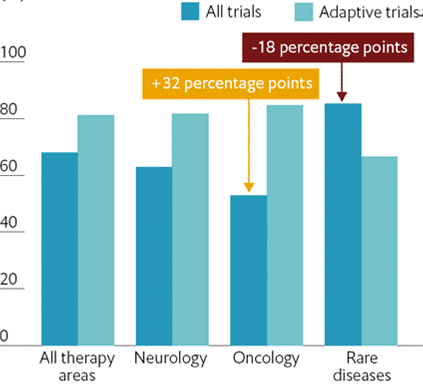
The reason for this success is because deploying decentralized trials and taking the study to the home of the patient is the epitome of patient-centricity. Not only will it help reduce the burden on patients, but it can also make patient’s lives easier and positively impact recruitment and retention efforts.
In addition, with the rise of accessible, “bring your own device” (BYOD) technologies, participants can overcome common clinical trial barriers, such as geography and technical proficiency. Operating on apps and devices that patients already know how to use can increase engagement and help minimize confusion, ultimately leading to reduced drop-out rates.
To make patient-centric technology more appealing to patients, sponsors and clinicians must present their patients with a unified package of tools that are intuitive, accessible, and robust. Nick Darwall-Smith, VP of Technology at PAREXEL stated, “When a patient looks at whatever they’ve been asked to use, whether it’s an app on a mobile device, an ECG device or a wearable glucose monitor, it has to be clear right away what they need to do. They shouldn’t have to take a training course to know how to use it.”
Decentralized Trials Provide Diverse Representation
As clinical trials adopt more patient-centric technology, it’s important to remember how that affects participants in various socioeconomic settings. Prior to the pandemic, most clinical trials still required patients to attend doctor visits in-person—sometimes multiple times a week. They also relied on patients keeping paper diaries documenting their symptoms and wellbeing. Additionally, recruitment was typically conducted via primary care physicians.
As a result, people with hourly wage jobs that offer little paid time off and single parents living in rural towns had bigger challenges with meeting these requirements. Arranging child care or accessing clinical research centers creates a burden that is just too high, despite the overwhelming good that clinical research could do to patients’ health.
By increasing the use of decentralized solutions, instead of depending on in-person visits or faulty paper processes, sponsors can reach previously unattainable patients and achieve more diverse representation. The way diseases manifest and drugs react can vary greatly by race, gender, and ethnicity. In addition to being a moral imperative, diversity helps treatments be more precise and take into account the genetic differences in people.
Although clinical trials have sometimes ignored the barriers that preclude minorities and those living in rural or disadvantaged communities from participating in clinical research, decentralized trials are breaking those barriers. Thanks to patient-centric mobile apps, remote monitoring solutions, and communication technologies, clinical researchers can check-in with patients as often as needed.
And they can do so without requiring arduous in-person visits. Not only does decentralization ensure better access to participants across geography, race, sex, and age, it also provides data necessary to understanding dosing, efficacy, and safety across a broader range of patients.
However, as clinicians increase access to wider populations, they should thoughtfully consider the “digital divide” that often plagues lower-income areas. When offering decentralized solutions, clinicians should not only enable participants to use the technology they already own whenever possible but also ensure that the tools can be properly provisioned to those who might not have access to necessary technologies.
Decentralized Trials Reach A Better Connected Generation
Not only do decentralized trials help clinicians and sponsors reach a wider population of participants, but they also strengthen engagement with more technology-savvy generations. According to a survey conducted by the Kaiser Family Foundation, nearly half of millennials (45%) don’t have a regular primary care physician compared to the 85% of people aged 50 – 64 and 88% of those aged 65 or above that do. These numbers can present a problem for clinical trials because primary care doctors are the most heavily relied-on source for recruiting clinical trial participants.
A decentralized trial that is unbound by geography and using tactics like digital advertising to appeal to potential patients could help solve the primary care problem and connect better with the largest living adult generation.
Once millennials have been recruited to a trial, sponsors are struggling to engage with a generation that relies heavily on mobile solutions for everything from work to personal banking and shopping. Decentralized solutions leverage tools and technologies that millennials and younger generations are more accustomed to using, which makes them more likely to participate. E-signatures, online forms, and wearables are much commonly used by millennials and younger generations in their daily lives, making it a preferred method for data collection instead of in-person visits and paper logs.
The Road Ahead
Decentralization can’t completely eradicate the issues surrounding poor recruitment and high drop-out rates. But as more clinical trials embrace technology that supports hybrid and virtual solutions, they are opening the doors for a more diverse and engaged participation pool.
Inevitably, some trials will require in-person visits for biopsies, radiology procedures, and other endpoints that require professional evaluations. But clinicians and sponsors can begin thinking critically about what areas of their studies might benefit from patient-centric decentralized technologies. They can start by asking themselves what the minimum number of onsite visits is needed to operate the trial and which visits can be supplemented with telehealth options. From there, they can determine how much data they want to collect through remote monitoring and connected devices.
Finally, conducting a decentralized trial isn’t just about having the right solutions with patient-facing technology. To truly be successful, the trial needs a unified platform that can also support and adhere to data privacy, compliance, and seamlessly integrate your remote or hybrid solutions.
Medrio’s technology was designed with the patient in mind. Our flexible, unified solutions capture data digitally via our ePRO, eConsent, and Direct Data Capture solutions and integrate automatically with our fully-compliant EDC. We also understand that it’s not just about using the right technology. That’s why our team of experts is available 24/7 to help navigate any of your decentralization needs.
References:
1 The Economist Intelligence Unit; The Innovation Imperative: The Future of Drug Development Part I: Research Methods and Findings; Commissioned by Parexel
